The light on the clinic’s ceiling flickers softly, like a tired star. Beneath it, a man in his forties stares at the small, moon-white device resting in the surgeon’s gloved hand. It’s not much bigger than a matchbox, smooth and quiet, with threads of tubing that look almost fragile. But this little box is something else entirely. It is, for the first time in human history, a cyborg pancreas—part flesh, part machine, and all hope.
Outside, the city moves in its usual rhythm: cars sigh past, coffee machines hiss, glucose levels spike and crash in millions of bodies. But in this room, time narrows to a single point. The man thinks of nights spent trembling from low blood sugar, days ruled by carb counts and finger-prick tests, vacations and dinners and first dates shaped around an invisible, relentless illness. And now, for the first time since his diagnosis, he is being offered a different kind of life. Not a cure, exactly. Something stranger. A partnership: human and machine, working together beneath his skin.
When Your Body Outsources a Job
Diabetes, for all the high-tech meters and glossy brochures, is a very old heartbreak. At its core, it’s a story about a single organ that can’t keep its promise. The pancreas, buried quietly behind your stomach, is supposed to sense sugar drifting in your bloodstream and release insulin with the grace of a perfectly tuned orchestra. Too much sugar, and the music turns to noise. Too little, and the lights go out.
For people with type 1 diabetes, the pancreas simply stops playing altogether; the insulin-producing beta cells have been destroyed, usually by the body’s own immune system. Type 2 diabetes often begins as a quieter rebellion: the body grows resistant to insulin, the pancreas works overtime, then slowly burns out. Different paths, same destination—glucose mismanagement, organ damage, endless vigilance.
Modern medicine has tried, for decades, to fill in for this missing organ. Syringes, pens, insulin pumps, continuous glucose monitors glued to the back of the arm, apps that prod and warn from the privacy of a pocket. Thousands of tiny decisions made each year: Should I eat this? How many units? Am I dropping? Am I safe to drive? Is this headache just a headache?
We’ve been building better tools, sharper data, smarter algorithms. But all of it, no matter how sleek, is still work. The human being has to be the boss, the mathematician, the night watchman. A cyborg pancreas asks a radical question: what if the body could outsource that job again—this time not back to a failing organ, but to a device built to share the load?
The Quiet Revolution Under the Skin
To understand how radical this is, you have to picture what’s actually happening beneath the skin. The first cyborg pancreas isn’t a single magic gadget; it’s a conversation between biology and engineering. Think of it as a small, implanted ecosystem.
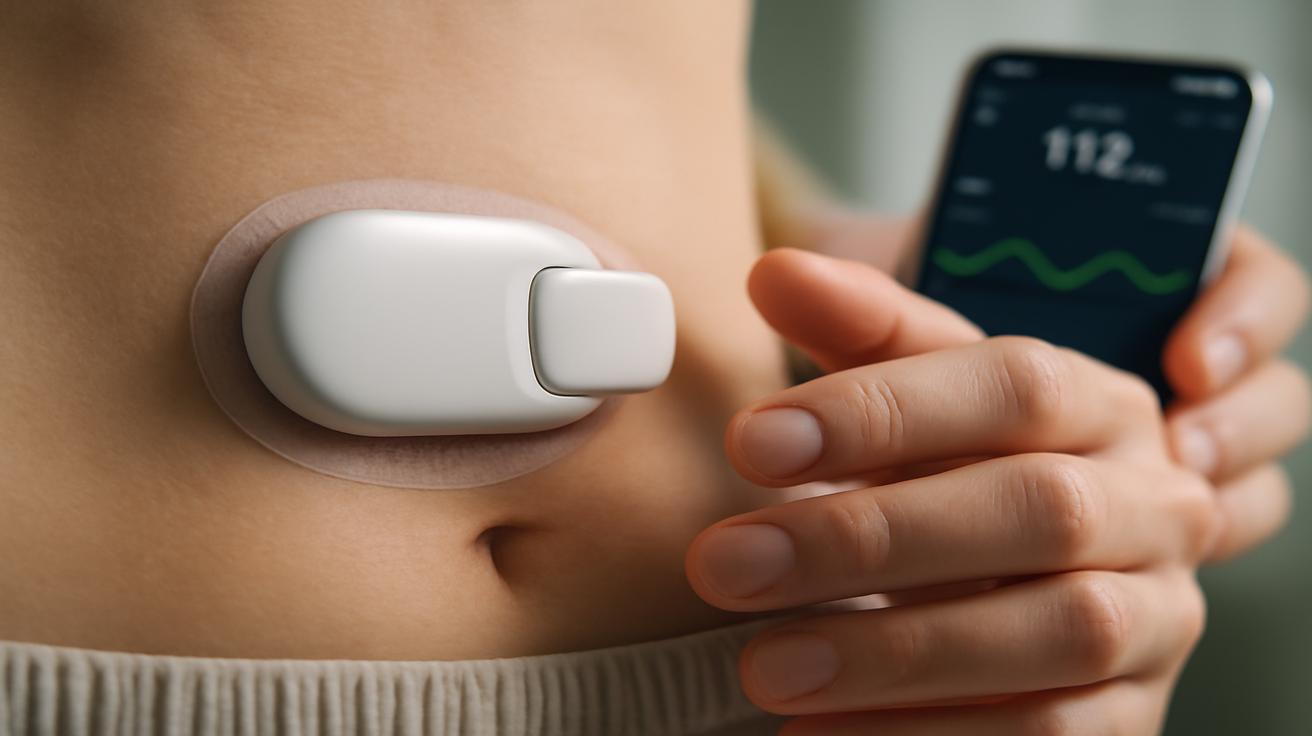
Inside the body, thin sensors listen to the ebb and flow of glucose in the bloodstream or interstitial fluid. They don’t just take a reading once in a while; they pay attention, the way a parent listens for a child turning in their sleep. Every few minutes—sometimes every few seconds—they whisper data back to a processor, a tiny brain built from silicon instead of cells.
This brain runs algorithms that would make a professional juggler proud. It weighs your recent meals, the trend of your sugar levels, how your body usually behaves at 3 p.m. versus 3 a.m., maybe even how active you’ve been. Then, nearly instantly, it decides: more insulin, less insulin, or—when the design allows—hormones that can nudge glucose upward if you’re dipping too low.
In response, a pump delivers just the right amount of insulin, drop by microscopic drop. No beeping, no tubes snaking out of your clothing, no need to pull out a phone in the middle of dinner to tap in numbers. To the outside world, nothing looks different. But inside, a new partnership is humming along, quietly bending the arc of the disease.
That’s what makes this first generation of cyborg pancreas devices so stunning: not that they exist, but that they try to restore something we lost—the feeling that the body can take care of itself again, at least a little.
From Finger Pricks to Feedback Loops
The road to this moment is paved with lancets and test strips. For years, managing diabetes was like trying to steer a car while only being allowed to open your eyes a few times a day. You’d prick your finger, test a droplet of blood, get a number, and then guess: should I correct with insulin? Should I eat? Should I wait?
Then came continuous glucose monitors (CGMs), those tiny sensors that live under the skin and send readings every few minutes. For many people, it was the first time they could see their glucose move like a living creature rather than a handful of disconnected snapshots. A bowl of pasta didn’t just “raise blood sugar”—you could watch it climb. A late-night walk didn’t just “lower your levels”—you saw the slope descending, a gentle ramp into safer territory.
Insulin pumps followed a similar path: at first clunky, then smarter, then smaller, dispensing insulin in a steady drip instead of in jolting injections. For some users, this meant tighter control and more freedom—no more bringing a pharmacy’s worth of supplies to every outing, no more turning a restaurant table into a minor medical scene.
The breakthrough came when these two devices started talking to each other. Closed-loop systems—nicknamed “artificial pancreas” setups—used CGM readings to automatically adjust insulin delivery. At first, the human still had to announce meals and calibrate regularly. But little by little, the burden eased. Some people got so comfortable that they hacked their own systems before regulators caught up, building DIY closed loops to let the machines handle more of the work.
The cyborg pancreas is the next step: move more of that intelligence, more of that feedback loop, inside the body. Reduce the surface clutter. Cut down the visible hardware. Turn a life-or-death chore into an invisible rhythm.
What Makes a Pancreas “Cyborg”?
The word “cyborg” can feel like it belongs in a neon-lit sci-fi alley, not in a medical journal. But stripped of the drama, it simply describes a hybrid: a living organism enhanced or supported by machines. A cochlear implant making sense of sound. A pacemaker pacing a faltering heart. A neurostimulator quieting the storms of Parkinson’s disease.
The first cyborg pancreas fits right into this lineage. It is built on three pillars:
- Sensing: It constantly measures glucose in the body, not every few hours, but continuously.
- Deciding: It uses algorithms—trained on data, refined through trials—to determine how much insulin (and in some designs, other hormones) to release.
- Acting: It delivers that dose automatically, without the wearer needing to calculate, tap, or inject.
This creates a feedback loop, a cybernetic system: the body provides data, the device interprets, the device acts on the body, and the cycle repeats. With each loop, your glucose stays in a narrower, safer range. Fewer spikes that leave you wrung out and wired, fewer crashes that turn your hands to jelly and your thoughts to fog.
And there’s another layer here, quieter but profound: the emotional feedback. Over time, as the device proves itself, your trust in it grows. You might sleep deeper. You might say yes to a hike, a late meal, or a long drive you would have once avoided. Your identity, once braided tightly with the rituals of diabetes, starts to fray and restitch itself into something new: no longer just a patient, but a collaborator with your own internal technology.
The Texture of Life With a Machine Pancreas
To really grasp what this means, imagine an ordinary day, reframed. You wake up not to an alarm reminding you to check your sugar, but to quiet. The device has watched over you all night, nudging you away from lows, trimming back highs. The dull headache you used to accept as “normal” morning is gone.
You step into the kitchen. There’s coffee, maybe toast, maybe something sweet that once required a careful mental algebra problem: carbs, insulin on board, current level, the timing of the spike. Now, you still think about food—but the background anxiety has softened. You tap in that you’re eating, if your system needs that hint, or you simply eat and let the loop do its job, if it’s advanced enough. You still carry backup supplies; this is diabetes, after all, and redundancy is survival. But the calculations are less frantic, less constant.
At work, you sit through a long meeting without secretly watching your glucose graph every few minutes. On a walk after lunch, the device trims back your insulin to avoid a crash. Later, as evening draws in, you don’t have to build your entire schedule around a perfect pre-bed reading. You know your tiny internal collaborator is wide awake, even when you’re not.
None of this means perfection. Devices fail. Sensors peel off. Algorithms make mistakes. But the baseline has shifted. The spikes are less violent, the crashes less frequent. The days feel less like tightrope walking and more like ambling along a trail that, while not perfectly smooth, is at least not made of glass and knives.
A Glimpse at the Tech Beneath the Hope
For those who like to peek under the hood, the first cyborg pancreas devices are a careful layering of hardware and software, biology and design. Here’s a simplified view of how their main elements compare with older approaches:
| Feature | Traditional Diabetes Management | Cyborg Pancreas Approach |
|---|---|---|
| Glucose Monitoring | Finger-stick tests a few times per day | Continuous internal or semi-internal sensing |
| Insulin Delivery | Manual injections or basic pumps | Algorithm-guided microdoses, automated |
| Decision Making | User calculates and adjusts throughout the day | On-board processor analyzes trends and acts |
| Visibility | External devices, visible infusion sets | Partially or fully implanted, mostly invisible |
| User Burden | High—constant attention, frequent decisions | Reduced—oversight instead of micromanagement |
That last row might be the most important of all. Diabetes has always demanded attention as a tax on time, energy, and mental space. A cyborg pancreas doesn’t erase that tax, but it negotiates it down.
Ethics, Access, and the Uneasy Future
Of course, every technological miracle arrives carrying its own shadows. The first cyborg pancreas is no different. For one thing, it is unlikely to be cheap. The engineering, the clinical trials, the regulatory pathways, the manufacturing—all of it adds up. Many of the people who could most transform their lives with this technology may find it resting behind a wall of cost, geography, or insurance politics.
There are also questions of autonomy and control. Who owns the data streaming from this device, data that maps your body second by second? How transparent are the algorithms deciding your insulin doses? Can you adjust them, or are you forced to trust a black box? When a device becomes part of your body’s ongoing survival, that relationship becomes deeply intimate. Trust has to be earned, and it has to be protected.
And then there’s the identity shift. For some, becoming a “cyborg” is empowering, even exhilarating. It feels like a reclamation of power from an illness that has taken so much. For others, the idea of merging so deeply with a machine stirs unease. Will they still feel fully themselves? Will others treat them differently once they know a device is quietly co-running their metabolism?
But woven through these hard questions is something gentler: the recognition that humans and tools have always evolved together. Spears and glasses, vaccines and smartphones—we change the world, and in turn, the tools we’ve made change us. The first cyborg pancreas is simply a more intimate step in a very long dance.
Beyond the First Implant: What Comes Next?
This first device is a prototype in the grand narrative of medicine: a beginning, not an endpoint. Future versions might better mimic the true pancreas, pairing insulin with its hormonal counterpart glucagon, or even integrating other metabolic hormones to soften the edges of mealtime spikes.
Materials will evolve too. Biocompatible coatings will aim to quiet the immune system’s suspicion, allowing sensors to last longer without being walled off by scar tissue. Smaller batteries, or clever ways to harvest energy from the body’s own motion, could shrink these devices further, making them easier to implant and live with.
We may also see a smoother integration between devices and living cells. Some research teams are working on bioartificial pancreases—tiny capsules containing real insulin-producing cells, shielded from immune attack yet still able to sense glucose and respond. Marrying those cells with smart delivery systems and algorithms could create hybrid solutions that blur the line between transplant and technology even more.
On a social level, if these devices prove themselves and become more widespread, our cultural picture of diabetes will shift. Schools, workplaces, and families may think less about immediate crises and more about long-term support. A teenager with a cyborg pancreas might go to sleepovers or backpacking trips with less worry. A parent may stop waking every few hours to check on a child’s reading, surrendering some of that fear to the steady hands of a well-tested algorithm.
A Radical Path, but Not a Lonely One
In that clinic, under that flickering light, the man eventually nods. Yes, he says. Let’s do it.
He doesn’t do it because he worships technology. He does it because he is tired. Tired of counting. Tired of calculating. Tired of having every joy negotiated through the stern language of milligrams per deciliter. He does it because he wants a little more quiet in his mind, a little more trust in his own body, however mediated by metal and code it may now be.
The first cyborg pancreas is not a cure. It does not erase the years lived with diabetes or the complications already seeded in nerves and vessels. It won’t work for everyone, and it won’t be perfect even for those who get it. But it is a door, and it opens onto a path that once seemed almost unthinkable: a future where chronic illness is not an endless manual chore, but a collaborative process between our cells and our machines.
We like to imagine radical change as sudden—a bolt of lightning, a single breakthrough, a moment that divides “before” from “after.” In truth, it’s often quieter. A small device rests in a surgeon’s hand. A person signs a form with a trembling signature. Somewhere beneath the ribs, biology makes space for circuitry. And inside that tiny space, our long, complicated relationship with our own bodies takes another step forward.
In the years to come, more people will carry these cyborg organs. They’ll walk through crowds, sit in classrooms, stand in checkout lines, unseen and unremarkable. Yet inside them, a quiet rebellion will be underway—against the inevitability of damage, against the tyranny of constant vigilance, against the idea that being ill means being forever at war with yourself.
The first cyborg pancreas does not announce this revolution with fanfare. It hums softly under the skin, counting, adjusting, caring in its own machine-like way. It is not human, but it exists to honor something deeply human: the desire to live a life shaped more by choice than by disease.
FAQ
Is a cyborg pancreas the same as an “artificial pancreas” system?
They are closely related ideas. Many artificial pancreas systems are external—pairing a continuous glucose monitor with an insulin pump and an algorithm. A cyborg pancreas takes that concept further by moving more of the sensing and dosing hardware inside the body, making the system more integrated and less visible.
Does a cyborg pancreas cure diabetes?
No. It does not restore the body’s own insulin-producing cells or stop the underlying autoimmune or metabolic processes. Instead, it manages blood sugar more automatically and precisely, reducing the daily burden and potentially lowering the risk of long-term complications.
Who might be eligible for a device like this?
Eligibility depends on specific device designs and regulatory approvals, but early candidates are often people with type 1 diabetes who struggle with severe highs and lows, or who have difficulty maintaining safe glucose control despite current technology and best efforts.
Are there risks to having a cyborg pancreas implanted?
Yes. As with any implant, there are risks of infection, device malfunction, immune reactions, and the need for surgical procedures to place or replace the system. These risks are weighed against the potential benefits in clinical trials and medical consultations.
Will people still need to think about food and exercise with a cyborg pancreas?
Almost certainly, yes. While the device can automate much of the fine-tuning of insulin delivery, major changes in eating, activity, or illness will still matter. The goal is not to make diabetes vanish, but to shift from constant micromanagement to a shared responsibility between person and device.








Leave a Comment