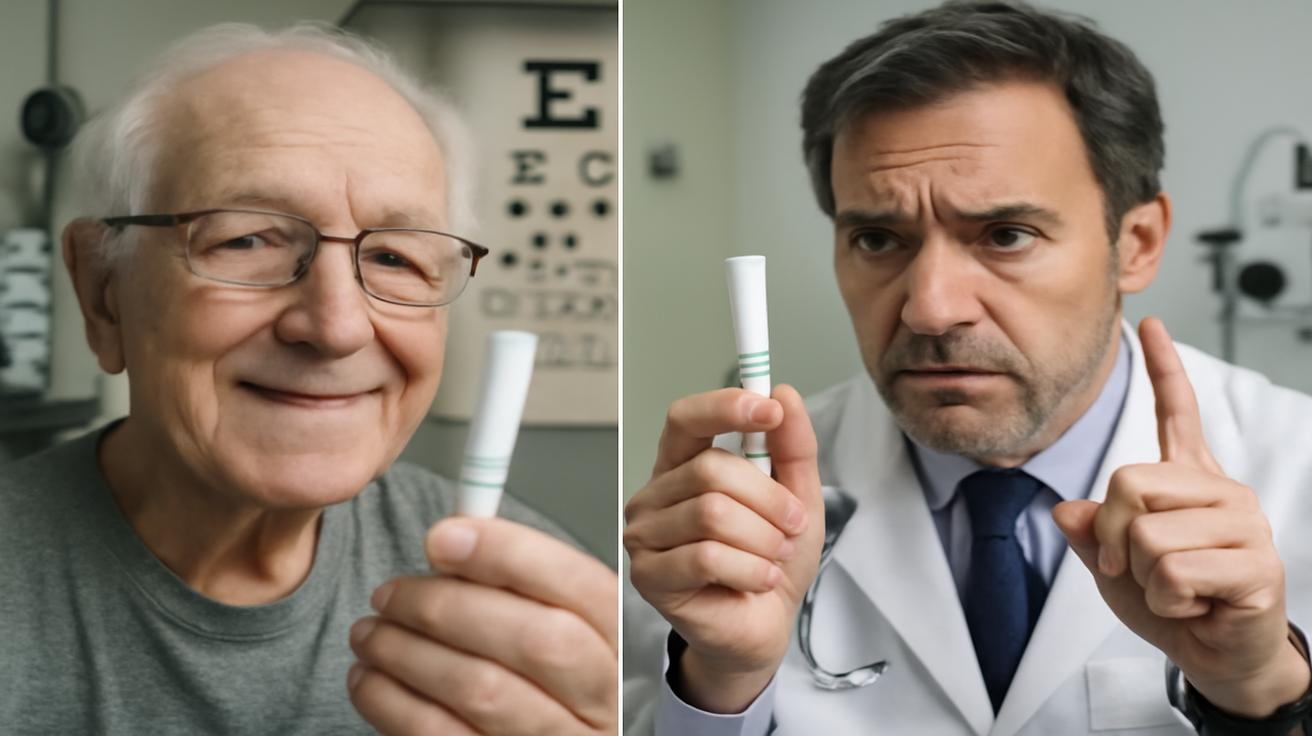
The woman in the blue scarf holds the tiny vial between her fingers as if it were a living thing. In the fluorescent hush of the exam room, she hesitates, feeling its cool glass against her skin. For the first time in years, she can see the outline of her own reflection in the metal counter—the pale blur of her face, the ghost of her features. Not enough to read a label or recognize her granddaughter across a room, but enough to remember what it meant to look out at the world instead of only imagining it. Her doctor nods, watching closely. “When you’re ready,” he says. She twists the cap, leans back, and lets a single, trembling drop of the new gel fall onto her eye.
The Promise Inside a Drop
Stories like hers have been traveling faster than any press release. Across waiting rooms and online forums, whispered between neighbors over garden fences, they follow a simple arc: a gel that isn’t a drug in the traditional sense, that doesn’t require lasers or scalpels, that is dabbed gently into the eye—and sight, long faded or dangerously clouded, begins to return.
In an age of incremental medical change, the very idea feels almost indecently hopeful. This so‑called “miracle eye gel” has been described as a kind of tissue whisperer, a clear, viscous liquid that carries molecules designed to coax damaged cells back toward function. It’s not magic. It’s chemistry and biology and decades of research braided together. But to the people leaning over sinks and bathroom mirrors, hands shaking with hope, it feels very close to a miracle.
They are the ones whose world has been shrinking—first the printed page, then street signs, then the faces they love. Many have already been told they’re not good candidates for conventional surgery, or that additional operations would be too risky. Some are frightened of invasive procedures. Others live far from the gleaming towers where delicate retinal surgeries are performed and follow-up appointments require hours of travel and a full day’s wages. For them, the possibility of healing in the quiet privacy of their homes is almost unbearably alluring.
But step outside that examining room and the story tangles. The same gel that patients describe in reverent tones is, in the eyes of many experts, a dangerous shortcut around hard-earned medical safeguards. To them, it is not a miracle but a high‑stakes experiment being run on people who have few options and fewer protections.
A Gel at the Edge of Medicine
The eye gel itself looks unremarkable—clear, slightly thicker than tears, held in small sterile vials. Inside, however, is a dense universe of design: a blend of biocompatible polymers, growth-factor mimics, and tiny messenger molecules meant to nudge certain cells into repairing or replacing damaged tissue. The exact recipe varies among developers, but the core idea is surprisingly simple: instead of cutting away the problem or installing an artificial solution, persuade the body to fix its own vision from within.
Some versions aim to regenerate the cornea, the transparent front layer of the eye that can become scarred after injuries or infections. Others claim to smooth out the stiffened lens proteins that cause cataracts, allowing light to pass cleanly again. A few experimental formulas whisper of even more daring goals: encouraging retinal cells to regrow in the back of the eye, where disease has gnawed away at the light-sensitive tissue that translates photons into sight.
The science is not born from nowhere. Researchers have been exploring regenerative medicine and gene-modulating therapies for the eye for years. In petri dishes and animal models, some of these approaches have worked astonishingly well. Mice with damaged corneas have seen tissue clear and smooth. Dogs with certain inherited conditions have regained measurable vision after targeted treatments. The eye, self-contained and accessible, has long tempted innovators.
What’s new is how quickly some of these ideas have leapt from lab bench to bedside table. In a handful of private clinics and pilot programs, patients are already sliding these gels into their eyes—not as part of tightly controlled trials, but as paying participants in what critics call an uncontrolled human experiment.
Stories of Sudden Clarity
In one coastal town, a former fisherman describes the first morning after using the gel. For years, the harbor outside his window had been no more than a milky smear of grays and whites. That day, he swears, he could make out the individual lines of moored boats, the sharp edge of a gull’s wing as it folded against the sky. “It was like someone turned the dial,” he says. “Not back to twenty, but back to possible.”
A retired teacher in her seventies, who had been told her cataracts were too advanced for safe surgery due to other health conditions, tells a similar tale. After weeks of carefully applying the gel twice a day, she says she can once again read the large-print novels that had sat untouched on her bedside table. “I still use a magnifier,” she admits, “but it’s my eyes doing the work again. Not just the tools.”
The clinic that dispensed her gel proudly catalogues such stories, collecting testimonials like bright shells along a beach. Their website speaks of “restored independence” and “non-surgical rejuvenation of sight.” Photos show patients smiling in sunlit parks, eyes crinkling with apparent gratitude. The language washes over the screen in soothing blues and greens, emphasizing “natural healing,” “biologic renewal,” and “avoiding risky procedures.”
For those who have never sat in the dim dread of an ophthalmologist’s office listening to the soft click of failed eye charts, the appeal might be hard to grasp. But when your world has blurred and narrowed, when every new smudge of vision feels like another door closing, even a hint of recovery can feel like oxygen. The gel is sold not just as a product, but as a rescue line.
The Fine Print in Fading Light
Somewhere near the bottom of the clinic’s informational handout, however, the tone changes. In the smaller paragraphs, in careful legal phrasing, words like “experimental,” “not yet fully evaluated,” and “individual results may vary” begin to appear. It is in these hushed fonts that the concerns of many eye specialists take root.
Most approved eye treatments, especially those that claim to alter tissue or cellular behavior, are the product of years—often decades—of stepwise testing. First in animals, then in carefully monitored human trials with control groups, placebo comparisons, and rigorous follow-up to catch late-emerging side effects. Data is collected not just on what improves, but on what goes wrong: infections, scarring, inflammatory damage, unexpected changes in pressure or shape inside the eye.
Many forms of this new gel have skipped or only partially completed that journey. Instead of entering the slow and expensive funnel of large-scale clinical trials, some versions are offered under therapeutic loopholes or in jurisdictions where oversight is patchy. Patients qualify by having a credit card, not by being randomly selected for a research cohort.
Critics argue that the consent forms—those lines of dense legal strategy printed in faint ink—cannot possibly convey the risks of meddling with one of the most intricate sensory organs humans possess. What, they ask, happens if regenerative signals are too strong or misdirected, spurring abnormal growth, new opacity, or even tumors? What if the gel triggers subtle inflammation that scars over months, undoing any initial gains and leaving the patient worse off?
Some ophthalmologists, accustomed to delicately precise surgical interventions, bristle at the gel’s marketing language. To them, “no major surgery” does not automatically mean “safer.” One surgeon points out that an expertly performed cataract operation, though invasive, has success and safety statistics polished over millions of procedures and decades of refinement. “We know exactly what we’re doing in there,” she says. “With this gel, we’re asking the eye to rewrite itself from the inside and then hoping it follows the script.”
Hope, Money, and a Narrow Window
The people most drawn to the gel often occupy a particular corner of the medical landscape. They are older, sometimes managing multiple illnesses. They may live on fixed incomes or far from urban medical centers. Many have been turned away from traditional surgical fixes or told that further interventions carry substantial risk. Others cannot afford time off work to recover from operations, or lack the support network needed to navigate a complex recovery.
For them, the decision to try the gel is rarely made in the abstract. It is made in dim kitchens over stacks of medical bills, in the long quiet after another appointment ends with “There’s not much more we can do.” In that space, a promise of regained sight for a fraction of the cost of surgery—and without hospital stays or the fear of the operating room—can feel irresistible.
Cost, of course, is part of the picture. These gels are not cheap. A full course may run into the thousands of dollars, and because many are not officially approved as standard treatments, insurance coverage is sporadic or non-existent. Patients are paying mostly out-of-pocket, betting savings and sometimes family support on a therapy that may or may not work and may or may not be safe long-term.
Yet the math they do in their heads is not only financial. How much is a year of clearer sight worth? How about a month? How do you quantify the first time in years you can see your own eyes in the mirror or read a grandchild’s birthday card without help?
What We Know — and What We Don’t
Beneath the swirl of marketing, caution, and personal testimony, there are some early data points. Small-scale pilot studies, some published and others circulating quietly among researchers, suggest that certain gel formulations can indeed improve corneal clarity or slightly reduce lens clouding in specific patient groups. In controlled settings, under careful monitoring, some participants have experienced measurable gains in visual acuity over weeks or months.
In these studies, the gels are applied under strict protocols. Patients are screened for preexisting conditions, followed up regularly, and examined with imaging tools that can detect subtle changes in tissue and intraocular pressure. Side effects such as temporary irritation, redness, or fluctuating vision are common, but often mild. The big questions—What happens after a year? Five years? Ten?—linger, unanswered.
Outside that controlled environment, however, the picture fragments. People get hold of gels or copycat products from less regulated sources, sometimes even compounded mixes that mimic the original concept without the same quality controls. Dosing schedules become casual guesses. Follow-up is haphazard. When something goes wrong—a sudden spike in intraocular pressure, a bout of painful inflammation, a new haze creeping across the field of vision—it may never be connected, on paper, to the gel at all.
Regulators face a dilemma. The drive to foster innovation, especially for conditions that lack good options, must be balanced against the duty to prevent harm. Move too slowly, and you strand desperate patients in limbo, blocking access to potential breakthroughs. Move too fast, and you risk enshrining hope as evidence and anecdote as data.
| Aspect | Miracle Eye Gel (Early Versions) | Traditional Eye Surgery |
|---|---|---|
| Approach | Non-surgical, regenerative or tissue-modulating drops/gel | Physical removal or replacement of damaged tissue |
| Evidence Base | Limited early studies; heavy reliance on patient stories | Decades of large clinical trials and outcomes data |
| Risks | Unknown long-term effects, potential inflammation or scarring | Surgical complications, infection, anesthesia risks |
| Accessibility | Can be used at home; often expensive and not insured | Requires operating room and specialists; variable insurance coverage |
| Regulation | Mixed; some products in gray zones or loosely regulated clinics | Tightly regulated procedures and devices |
The Ethics of an Unfinished Cure
Ethicists who study emerging medical technologies often return to a simple but thorny question: At what point does offering an experimental therapy stop being compassionate and start being exploitative? The line is rarely bright. It shifts depending on context, urgency, and how honestly limitations and uncertainties are communicated.
With the eye gel, desperation is the silent partner in every conversation. When people feel they are running out of time—time with meaningful sight, time to watch a child grow, time to move through familiar streets unaided—they may be willing to overlook or minimize risks. That vulnerability creates fertile ground for both well-intentioned overpromising and outright predatory marketing.
Some clinics, to their credit, are trying to walk a narrower path. They require extensive informed-consent discussions, emphasize that results are not guaranteed, and encourage patients to continue seeing their regular eye specialists for objective monitoring. A few are even collecting structured data, hoping to contribute to the broader understanding of what the gel can and cannot do.
Still, the fundamental imbalance remains: the patient is betting their only pair of eyes on a treatment whose full story is not yet written.
Between Darkness and Dawn
If you step back from the drama and listen for the quieter undercurrents, something else emerges. The gel, in all its controversy, has pulled an uncomfortable truth into the light: modern eye care, for all its precision and success stories, still leaves many people behind.
There are the predictable gaps—rural areas without enough specialists, communities where cost and transport form an invisible but impenetrable barrier. There are also the less visible failures: patients who are medically “too complicated” for standard surgeries, whose coexisting illnesses make anesthesia or wound healing a frightening gamble. For them, a drop of gel that whispers “maybe” into the silence of “no more options” feels like an opening where none existed.
That doesn’t mean the gel, in its current form, is the answer. It might never be, at least not without substantial refinement and careful, transparent testing. Some versions may prove to help only in very specific cases. Others may reveal hidden harms when used at scale. Still others might quietly morph, through years of research and iteration, into something safer and more predictable—less miracle, more medicine.
But even if this generation of gels falls short of its promises, the hunger it has revealed will remain. A hunger not only for restored sight, but for treatments that respect the full context of people’s lives—where they live, what they can afford, what they are afraid of, and what they hope for when the lights begin to dim.
Back in the exam room, the woman in the blue scarf blinks against the first cool touch of the gel. The world doesn’t snap into focus; there is no cinematic moment of sudden clarity. Her vision remains a soft blur. The doctor checks her eye pressure, peers into the glowing circle of her pupil with practiced concentration. They agree to watch and wait. Outside, afternoon light spills across the parking lot, sharp and indifferent.
In the weeks ahead, she may see changes—or she may not. Her story might join the collection of quiet triumphs that clinics highlight on glossy brochures, or it might disappear into the vast, invisible archive of treatments that didn’t quite deliver. But for now, in this thin wedge of time between darkness and dawn, she has something she has not felt in years: the sense that the story of her sight is not completely finished.
In that fragile space, the miracle gel is both promise and provocation—reminding us not only of what science can do, but of how carefully we must move when the path forward is lit mostly by hope.
Frequently Asked Questions
Is this “miracle eye gel” an approved treatment?
In many places, early versions of these gels are not fully approved as standard treatments. Some are used in tightly controlled research settings, while others are offered through private clinics under regulatory loopholes. Approval status varies widely by country and region, so it is essential to ask your eye specialist and check with local health authorities.
Can the gel really restore sight without surgery?
Some patients report noticeable improvements, especially in conditions involving corneal clarity or early lens clouding. However, evidence is still limited, and results are highly variable. For advanced disease or complex retinal problems, current gels are unlikely to fully replace surgery or other established treatments.
What are the possible risks?
Known short-term risks include irritation, redness, fluctuating vision, and potential increases in eye pressure. The major concern is unknown long-term effects—such as scarring, chronic inflammation, or abnormal tissue growth—because large, long-term studies are still lacking.
Who might consider trying the gel?
It is sometimes considered by people who are poor candidates for conventional surgery, are deeply fearful of invasive procedures, or have limited access to surgical care. Anyone considering it should first have a thorough evaluation by a qualified ophthalmologist and discuss all established alternatives.
How can I protect myself from risky or fraudulent offers?
Be wary of bold guarantees, limited-time offers, or clinics that discourage second opinions. Ask whether the product has been evaluated in peer-reviewed clinical trials, who manufactures it, how complications are handled, and whether your regular eye doctor can be involved in monitoring your eyes throughout treatment.
Will future versions of these gels be safer and more effective?
It is possible. Regenerative and tissue-modulating eye therapies are active areas of research, and early results are encouraging in specific contexts. With rigorous testing, clearer regulations, and transparent reporting, future formulations may eventually become part of mainstream eye care.
What should I do if I’m losing vision now?
Start with a comprehensive eye exam from a trusted professional, ideally a board-certified ophthalmologist. Ask about all evidence-based options for your specific condition, including surgery, medications, lifestyle changes, assistive devices, and, if appropriate, carefully monitored clinical trials for new therapies. Above all, avoid making decisions based solely on testimonials or advertising when your sight is at stake.







Leave a Comment