The nurse dimmed the lights, and the room fell into a soft twilight hum of machines and whispered conversations. On the monitor, glowing lines traced the quiet rhythm of a life that had begun to revolve around scan results, blood tests, and the suspense between appointments. To the person in the chair—blanket pulled to the chin, plastic ID band loose around the wrist—cancer didn’t feel like a “cellular malfunction” or a “genetic error.” It felt like something cunning, something that had learned how to hide in plain sight.
That’s the thing about cancer. It doesn’t always behave like a foreign invader. It adapts. It disguises itself. It learns to slip past the watchful patrols of the body’s immune system like a familiar face blending into a crowd. For decades, that invisibility has been one of its greatest weapons. But a new wave of research is quietly rewriting this story—by doing something both simple and radical.
Instead of just trying to kill cancer cells from the outside, scientists are teaching the immune system how to see them. And they’re doing it by turning cancer’s best disguises into bright, unmistakable targets.
When the Body Forgets What the Enemy Looks Like
Every second, your body is scanning itself. Immune cells float through blood and tissues like neighborhood watch volunteers, checking IDs. They look for surface markers—tiny molecular name tags—on the outside of every cell. If something looks strange or wrong, they investigate. If it looks dangerous, they launch an attack.
Cancer finds its foothold in a crack in this system. It starts as a normal cell that goes slightly off-script. A mutation here, a broken control switch there. At first, those altered cells may still look mostly familiar. Their name tags aren’t screaming “danger” yet. So the immune system shrugs and moves on.
As cancer grows, it becomes a master of subtlety. It sheds or mutates the very markers that might attract attention. Some tumors lose the key molecules that T cells use to recognize them. Others actively produce signals that calm nearby immune cells, like handing out soothing messages that say, “Nothing to see here, move along.”
The immune system, powerful as it is, relies on visibility. It needs contrast: the clear difference between “me” and “not me.” When that difference blurs, cancer spreads silently.
So researchers began to ask a new kind of question: What if we stop chasing cancer’s shape-shifting tricks and instead force it into a corner? What if we can give those hidden tumor cells a new, unmistakable flag—something the immune system will never overlook?
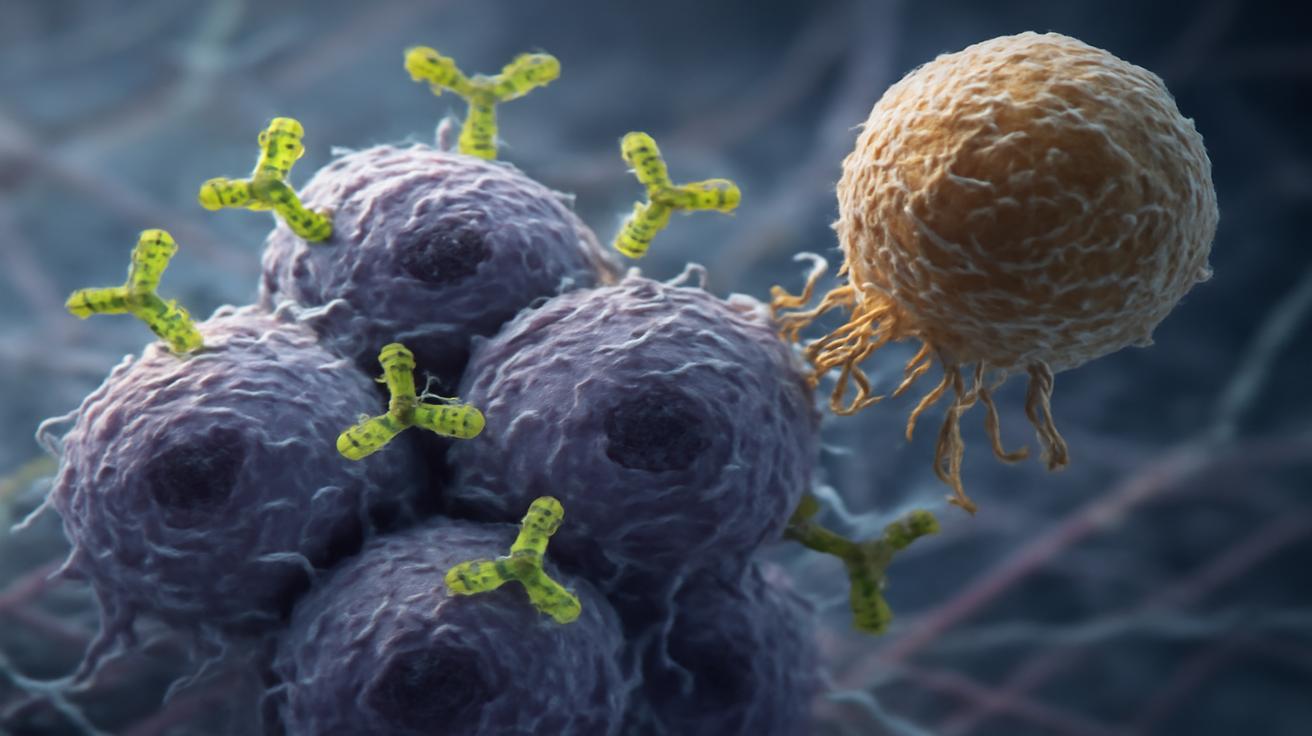
Painting a Target on Cancer Cells
Imagine walking into a crowded train station where you’re supposed to find a single person you’ve never met before. Now imagine everyone in the station is wearing the same gray coat. That’s the immune system trying to find early cancer cells in a sea of normal tissue.
Now change the rules. What if every cancer cell suddenly started wearing a bright red scarf?
This is the essence of a groundbreaking strategy now emerging from several research groups around the world: make cancer cells visibly different by decorating them with markers the immune system already knows and fears. Not with chemotherapy poisons. Not with random mutations. With clear, recognizable molecular “red flags.”
Some scientists are experimenting with small engineered molecules or nanoparticles that home in on cancer’s quirks—its overactive growth signals, overexpressed surface proteins, or metabolic oddities. Once they bind to a tumor cell, they carry with them a sort of fluorescent badge: a marker that screams, “I don’t belong here.”
Others are taking a more genetic approach, introducing safe, controlled changes into tumor cells that cause them to display completely new antigens—targets the immune system is primed to attack. It’s like forcing every misplaced cell to hold up a bright sign that says, “Hit me.”
In some experimental models, this involves coaxing cancer cells to express viral-like proteins on their surface. The immune system is exquisitely tuned to recognize viral signatures. By tricking tumors into “putting on” these viral masks, researchers are making them look contagious, dangerous, and impossible to ignore.
Suddenly, the gray-coat blur of the train station becomes a room full of red scarves. The watchful immune sentinels know exactly where to go.
A Silent Conversation Between Tumor and Immune System
There’s a quiet, biochemical language that flows between cancer cells and immune cells. Cytokines, receptors, checkpoints—these are the words and punctuation of an invisible conversation. For years, cancer has been winning this conversation, whispering: calm down, stand down, don’t look too closely.
Checkpoint inhibitor drugs were one of the first major successes in breaking that spell. Medications like anti-PD-1 or anti-CTLA-4 essentially tell exhausted T cells, “You’re allowed to fight again.” For some patients, the results have been nothing short of astonishing. Tumors melt away as dormant warriors snap awake.
But there’s a catch. To fight, T cells still need something to recognize. If the tumor has erased, hidden, or minimized its distinctive signs, unshackled T cells may find very little to attack. It’s like giving a soldier permission to enter the battlefield—but not handing them a map of where the enemy is.
That’s where this new “visibility” strategy takes on profound meaning. By forcing tumors to present bright, unnatural flags, scientists are handing the immune system that missing map.
Now imagine pairing these ideas: drugs that remove immune brakes, plus technologies that paint neon targets on the cancer. The dialogue changes. The tumor is no longer whispering; it’s being forcibly subtitled in bold letters the immune system can read.
Some early lab and animal studies suggest that when tumors are made more visible, even previously “cold” cancers—those with few immune cells nearby—can turn “hot,” swarming with T cells, natural killer cells, and other immune players. The once-quiet tumor site becomes a scene of controlled chaos: a cleanup operation, a reclaiming of territory.
How Scientists Are Teaching the Immune System to See
In modern labs that hum with incubators and microscopes, this work can look deceptively ordinary. A researcher in a white coat pipettes a nearly invisible liquid into a tube. A set of cells spins in a centrifuge. Under a microscope, a cluster of glowing dots appears on a computer screen. But behind those dots is a stunning change in strategy.
Here are some of the key experimental approaches scientists are exploring to make cancer cells visible:
- Engineered “paint” molecules: Tiny, customizable proteins or nanoparticles are designed to bind only to cancer cells and drag immune-recognizable tags onto their surfaces.
- Viral mimicry: Tumor cells are coaxed to display safe versions of viral antigens, activating powerful immune responses usually reserved for infections.
- Synthetic antigens: Researchers introduce artificial markers that don’t normally exist in the body but can be pre-taught to the immune system, creating a custom lock-and-key.
- Metabolic tagging: Cancer’s unusual metabolism is used against it, feeding it special building blocks that become bright “labels” on its surface.
Each approach has the same underlying goal: stop arguing with cancer’s camouflage and instead change the uniform it’s wearing.
From Petri Dish to Patient: Where Things Stand
Right now, many of these visibility-enhancing techniques live in the realm of early research—mouse models, organoids, and carefully controlled lab settings. Human trials are only just beginning for some of the most promising approaches.
But the early signs are stirring. Some experimental therapies have shown that once tumors are tagged with highly visible markers, the immune system doesn’t just attack them more aggressively; it also seems to learn from the battle. That learning is crucial. It hints that, given the right introduction, the immune system can build a kind of memory of the cancer, reducing the risk that it will sneak back later in a new disguise.
Of course, there are challenges. Making cancer cells stand out without confusing the immune system into attacking healthy tissue is a delicate balance. Scientists are obsessively fine-tuning how specific these tags are, how long they last, and how deeply they penetrate into a tumor’s tangled core.
There are also practical questions: How will these strategies blend with current treatments? Can they be delivered through an IV drip, a localized injection, or even a pill? How will they behave inside the complex ecosystems of different cancers—lung, breast, brain, pancreas—each with its own landscape and defenses?
Yet, as researchers test, adjust, and test again, a picture is forming of a future where visibility is a central pillar of cancer therapy—standing alongside surgery, radiation, drugs, and cell-based therapies as a fundamental way we tip the balance in favor of the body.
A Closer Look: Making Cancer Stand Out
To see how radically this shifts our approach, it helps to compare traditional and emerging ideas side by side.
| Approach | Traditional Focus | New “Visibility” Focus |
|---|---|---|
| Chemotherapy | Directly kill fast-dividing cells, cancer and healthy alike | May be combined with tagging to expose surviving cells |
| Radiation | Damage DNA in a defined area to shrink tumors | Can increase antigen release, amplifying visibility tags |
| Immunotherapy (checkpoints) | Remove brakes from T cells so they can attack | Work better when tumors are brightly tagged as targets |
| Targeted drugs | Block specific growth signals inside cancer cells | May help reveal or enhance immune-visible markers |
| Visibility strategies | N/A – largely new category | Mark tumors with clear, recognizable flags for immune attack |
Seen in this light, “making cancer visible” isn’t a replacement for current tools. It’s a translator, turning a once-murky enemy into something the rest of our therapies—and our own biology—can understand and respond to more precisely.
The Emotional Weight of Being Seen
Behind every clinical trial ID and every fluorescent image of labeled tumor cells is a human being who has sat in that dim room, listening to the quiet beep of monitors, waiting for answers. For them, talk of antigens and nanoparticles is ultimately about something very simple and deeply human: the hope that their body will not be fooled again.
There is a particular kind of fear that comes after treatment, when scans are clear but uncertainty lingers. Has the cancer really gone? Or is it hiding somewhere the machines can’t see yet? Patients describe it as a ghost at the edge of their vision, a shadow that moves when they turn their head.
The notion that we might someday teach the immune system to keep a vigilant memory of cancer—to recognize even a few stray, newly visible cells and dispatch them quietly before they bloom into trouble—is more than a scientific goal. It’s emotional relief. It’s the difference between waiting for the other shoe to drop and knowing, deep in the body’s own defenses, that someone is still on watch.
Ask a person in remission what they want most, and many will say: time. More birthdays, more ordinary mornings, more conversations that have nothing to do with test results. Strategies that turn cancer from a master of disguise into a brightly marked intruder aren’t just technical advances; they are, in a way, gifts of time.
Nature’s Own Surveillance, Upgraded
There’s also something quietly poetic about this work. For all our advanced machines and synthetic molecules, what scientists are ultimately betting on is the wisdom of the body itself. Evolution has spent millions of years honing systems that can recognize pathogens, clear infections, and remember them for a lifetime. We are not building a new defense from scratch. We are upgrading, guiding, and refocusing one that already exists.
In a forest, when a single tree gets sick, chemical signals drift through the soil and air. Neighboring trees adjust, bolster their defenses, and sometimes even send support through networks of roots and fungi. It is a kind of community immunity, an ecosystem watching out for itself. In our tissues, the immune system is playing a similarly intricate game. The new visibility strategies give it clearer signals, clearer stories about where danger lies.
The ultimate vision is almost elegant in its simplicity: cancer cells that can’t hide, immune cells that can’t be easily fooled, and treatments that nudge this relationship instead of bulldozing it. A conversation between our cells and our therapies, rather than a war waged entirely from the outside.
A Future Where Cancer Has Nowhere to Hide
We are not there yet. These strategies are, for now, the work of early-stage trials and glowing microscope images in quiet lab buildings. There will be setbacks. Some tumors will resist even this. Safety questions will need artful answers. But step by step, study by study, the era of invisible tumors may be narrowing.
In the near future, a person newly diagnosed might hear a different kind of treatment plan: We’ll not only shrink your tumor, we’ll also mark any remaining cells so your immune system can find them. We’ll watch not just with scans, but with blood tests that look for the very labels we’ve taught your body to recognize. And long after your last infusion, your immune memory will still hold a sketch of the enemy’s face.
For the person in the reclining chair, blanket pulled close, that shift might not feel like a scientific revolution. It might feel like a quiet, profound reassurance: this time, my body will see what it’s up against.
Cancer has spent decades winning by playing a game of hide-and-seek. The new science is simple in its promise and bold in its execution: turn on the lights, mark the hiders, and let the body do what it was always meant to do—protect, remember, and heal.
Frequently Asked Questions
What does it mean to “make cancer cells visible” to the immune system?
It means adding or revealing clear molecular markers on cancer cells so that immune cells can easily recognize them as abnormal. These markers act like bright flags that signal, “attack me,” helping the immune system target tumors more effectively.
Is this the same as regular immunotherapy?
It’s related but distinct. Many current immunotherapies remove brakes from immune cells or boost their activity. Visibility strategies focus on the other side of the equation: changing the cancer cells so they’re easier for those immune cells to detect and attack.
Are these visibility-based treatments available now?
Most are still in preclinical research or early clinical trials. Some patients may access them only through carefully controlled studies. It will take time to fully understand their safety and effectiveness in different cancer types.
Could making cancer more visible cause the immune system to attack healthy cells?
That’s one of the main concerns researchers are working hard to avoid. The markers used to tag tumors are being designed to be as specific as possible to cancer cells, and early tests focus heavily on ensuring that healthy tissues are not mistakenly targeted.
Will this replace chemotherapy or radiation?
Probably not entirely. The most likely future is a combination approach, where visibility strategies work alongside existing treatments like chemotherapy, radiation, targeted drugs, and other immunotherapies to improve outcomes and reduce relapse.
How soon could this change standard cancer care?
Timelines are uncertain. For some cancers and specific strategies, meaningful results might appear in the next several years. Broad, routine clinical use will depend on successful trials, regulatory review, and long-term safety data.
What can patients do now if they’re interested in these new approaches?
Patients can talk with their oncology team about ongoing clinical trials and research centers exploring advanced immunotherapies. While not everyone will qualify for early studies, staying informed and connected to major cancer centers increases the chance of accessing emerging options when appropriate.








Leave a Comment